Question: We are thinking of going through an IVF, but we are concerned whether IVF babies have higher risk of abnormalities. We are also concerned that IVF babies are less healthy than their counter-parts and have shorter life-span, is it true?
Dr Helena’s answer:
Since the advent of IVF and Assisted Reproductive Technology, there had been constant debates on its safety and implication towards the health of the next generation. One of the biggest concerns is whether these fertility treatment will give rise to increase risk of congenital abnormalities in babies born as a result of IVF.
These debates and concerns are certainly valid. After all, the process of IVF in creating life outside human body in a Petri dish and then putting them back into the human body to let it grow into a baby is certainly mind blowing. How would we ensure that the doctors and scientists know what they are doing? And mind you, the IVF process were the results of many trials and errors. What makes you think the product (which is the baby), is not plagued with defects and errors?
Intensive research in the early years, and a thriving population that has now grown to more than 5 million IVF children worldwide, have reassured scientists, but they have not stopped studying and trying to improve the process.
Recent discoveries in epigenetics – the study of how environmental factors can affect gene activity, and how a person’s risk of getting chronic diseases is “programmed” into them before they are even born – have opened up new possibilities.
Much of today’s research stems from the Barker hypothesis, which proposes that birth weight may be linked to the likelihood of getting certain diseases. IVF babies are known to have lower average birth weights – even if the difference, at about 20-30 grams, is small. Scientists are now investigating whether IVF conception equates with more hospital admissions, and an increased risk of cardiovascular disease, high blood pressure and diabetes in later life. However, there is no good evidence as yet to suggest likewise
A recent study by the Human Fertilization and Embryology Authority, which linked 106, 381 HFEA register records from 1992-2008 to the UK’s National Registry of Childhood Tumours (NRCT) is one of the largest population-based linkage studies ever carried out. This study has found no association between ART and childhood cancer. This finding offers comfort to those patients facing the difficult decision about whether to undergo fertility treatment or not.
In 2012, scientists at the University of California, Los Angeles (UCLA) looked at birth defects among infants born both via IVF and conceived through natural means in California, which has the country’s highest rate of IVF use. They included babies born after IVF and other assisted reproductive treatments such as couples’ use of fertility-enhancing drugs and artificial insemination.
Among 4795 babies born after IVF and 46,025 infants who were conceived naturally, 3,463 babies had congenital birth defects. Even after controlling for factors that can affect such birth defects, such as mother’s age, and race, which can influence rates of genetic and environmentally driven developmental disorders, 9% of infants born after IVF had birth defects compared to 6.6% of babies who were conceived naturally. Overall, the babies born after IVF were 1.25 times more likely to be born with abnormalities. The researchers did not find a link between birth defects and other fertility treatments like artificial insemination or ovulation induction.
It’s possible that the higher rate of abnormalities with IVF is due in part to whatever was contributing to infertility in the first place, say the researchers. But some of the researchers’ view was the fact that an increase was not seen among babies conceived using artificial insemination or ovulation induction suggests that process of IVF itself, in which eggs are removed from a woman, fertilized in a dish with sperm and then allowed to develop into embryos, which are transplanted back into the womb, is the primary culprit.
However, another more recent study in 2016 by researchers from the University of Adelaide and the University of Melbourne. The study reviewed all assisted reproduction technologies carried out in South Australia over a 16-year period from 1986 to 2002.
This was linked to data on birth outcomes from the South Australian Birth Defects Register (SABDR). The register includes a record of all live births, stillbirths, terminations, birth weight and congenital defects. Birth defects were also followed up for five years. The researchers looked at the statistical link between maternal factors and birth defects, and compared this between babies either conceived naturally or by IVF and ICSI. The study found no statistically significant increase in birth defect. There was some suggestion by the press that this study suggest that IVF reduces the incidence of birth defect in women after the age of 40, but was refuted by the research group due to its misleading nature.
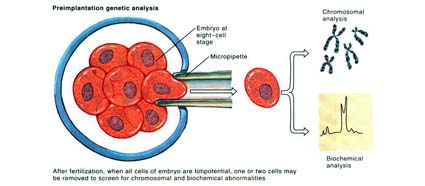
With the advent of Pre-implantation genetic screening (PGS) & Pre-implantation genetic diagnosis (PGD), which is fast gaining popularity, scientist can now screen embryos to exclude chromosomal and genetic abnormalities. The use of these technologies may further decrease the incidence of birth defect and congenital abnormalities associated with chromosomal defects or genetic issues. However, more long term data is required to support this hypotheses.
In conclusion, although there had been great hypothetical concerns about the risk of cancers & birth defects amongst babies born following Assisted Reproductive Technologies (ART), the actual link is difficult to establish due to many confounding factors such as parental age, the cause of infertility and etc. the actual incidence of childhood cancers and birth defects are small and should not be a great stumbling stone to those who are considering going through fertility treatment to have their babies